I took your comment to heart, and was wondering if I had remembered my oil experiences correctly.(I was not aware of the PAO formulation from ethylene gas)
Here's a blurb from Wiki/ReliablePlant/Noria Corp. on the PAOs(synthetics)--They specifically cite the low wet out.
You can envision this effect that PAOs have(further down in the article when it talks about the relative molecular size) by thinking of filling a bowl with, say golf balls(PAOs) Then fill a similar bowl with BBs(petroleum oils). the BBs will have much higher "surface contact" --ie. more BBs will be touching the bottom of the bowl than golf balls.
All this is really kind of moot, since most of us-just like you said wouldn't go the almost three times expense for these lubes
if nothing else, at least it's kinda interesting info.
Polyolefin surfaces are not effectively joined together by
solvent welding because they have excellent chemical resistance and are unaffected by common solvents. They can be adhesively bonded after surface treatment (they inherently have very low
surface energies and don't wet-out well (the process of being covered and filled with
resin)), and by some superglues (
cyanoacrylates) and reactive (meth)
acrylate glues.
[2] They are extremely inert chemically but exhibit decreased strength at lower and higher temperatures.
[3] As a result of this, thermal welding is a common bonding technique.
A more specific type of olefin is a
poly-alpha-olefin (or poly-α-olefin, sometimes abbreviated as
PAO), a polymer made by polymerizing an
alpha-olefin. An
alpha-olefin (or α-olefin) is an alkene where the carbon-carbon
double bond starts at the α-carbon atom, i.e. the double bond is between the #1 and #2 carbons in the
molecule. Alpha-olefins such as
1-hexene may be used as co-monomers to give a
alkyl branched
polymer (see
chemical structure below), although
1-decene is most commonly used for lubricant base stocks.
[1]
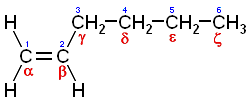
1-hexene, an example of an alpha-olefin
Many poly-alpha-olefins have flexible alkyl branching groups on every other carbon of their polymer backbone chain. These alkyl groups, which can shape themselves in numerous
conformations, make it very difficult for the polymer
molecules to align themselves up side-by-side in an orderly way. This results in lower contact
surface area between the
molecules and decreases the
intermolecular interactions between molecules.
[4] Therefore, many poly-alpha-olefins do not crystallize or solidify easily and are able to remain oily,
viscous liquids even at lower
temperatures.
[5] Low molecular weight poly-
alpha-olefins are useful as synthetic
lubricants such as
synthetic motor oils for vehicles and can be used over a wide temperature range
No disrespect, but the "not carbon based" is wrong. Of course they are carbon based. They are long hydrocarbon chains synthesized from ethelyne gas.
Rods and more importantly the bearing surfaces are metal, last time I looked.
There is no evidence here or anywhere else that what you are saying about "running out of the bearings" is true either. All engines are designed to retain small amounts of lube to aid start ups. Whether it's synthetic or mineral based makes no difference.
Back to the original poster-synths are better in every possible way. They are not even close to worth it though, in an older engine like a 2F. Maybe if you have a fresh perfect rebuild and you plan to run it 300K miles it would be but for your average good running but worn 2F, not worth it.

